The XV Collection: Auxin Transport — more a River Delta than a Stream
By Mark Estelle
The sessile lifestyle of plants is enabled by remarkable developmental plasticity. Plant form is affected by a wide range of environmental conditions from nutrient availability to herbivory, so two plants with the same genotype can appear quite different, depending on their environment. All plant organs and tissues are derived from dispersed stem cell populations called meristems. Individual meristems can act independently to a certain extent but are also controlled by systemic signals that coordinate and integrate their activities. The identity of these signals and how they function, has long been a subject of intense interest to plant biologists. The article that I chose to highlight for the PLOS Biology XV Collection is by Tom Bennett, Ottoline Leyser and colleagues and provides important new insight into the nature of systemic communication.

One of the best systems for understanding how plant development is coordinated is the behavior of meristems that are found in the axil of each leaf. These axillary meristems often persist in a dormant state, as buds, until such time as they are activated. For example, if the shoot tip is removed by herbivory or with a pair of scissors, bud dormancy is released, and a branch is produced. This phenomenon, called apical dominance, depends upon movement of the plant hormone auxin from the apex of the plant down through the stem. It is well known that auxin moves via a specific transport system called the polar auxin transport stream or PATS. PATS acts to rapidly move auxin through files of cells in the stem, specifically the xylem parenchyma and vascular cambium, from the plant apex down into the root system. Auxin transport requires one of a family of auxin cellular efflux carriers called the PIN-FORMED (PIN) proteins. Polar transport is the result of localization of the PIN1 transporter to the basal side of the cell.
Over the last decade, the Leyser group has performed elegant studies to explain how PATS regulates shoot branching. For bud activation to occur, a PATS must be established that transports auxin from the bud into the existing PATS in the stem. This appears to happen through a canalization process in which passive auxin transport from a source to a sink, upregulates and polarizes PIN transporters leading to formation of narrow polarized transport stream. Whether or not this happens depends on the source-sink relationship between the bud and the stem PATS. If auxin levels in the PATS are high, it will be a weak sink and transport from the bud will not occur. In contrast, if auxin levels are low, as is the case after decapitation, the stem PATS will be a strong sink, leading to establishment of a new PATS from the activating bud into the stem. It’s important to note that the PATS is sensitive to many other factors besides decapitation (a dramatic environmental stimulus) including light, nutrient availability and the genetic program. In this way the PATS functions as a central integrator of shoot branching control.
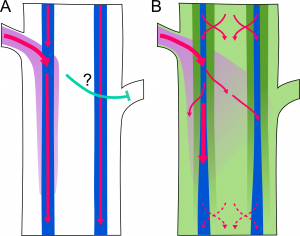
Although both experimental and mathematical modeling approaches support this model, it does not explain all aspects of branching regulation. For example, buds on opposite sides of the stem will transport auxin into different vascular bundles, yet still inhibit each other’s activation. To try to explain this sort of behavior, Bennett et al. explored the possibility that other auxin transport streams may exist in the stem. In particular, they investigated the role of other members of the PIN family in branching regulation. Their results indicate that auxin transport is much more complex than previously proposed. In addition to PIN1-based highly polar transport, PIN3, PIN4, and PIN7 contribute to widespread and less polar auxin transport, which they term “connective auxin transport” (CAT). Modeling studies demonstrate that CAT provides important local information that helps to coordinate the behavior of axillary meristems that are close together.
This work is extremely satisfying because it takes us beyond the simplistic view of polar auxin transport that has dominated the field for decades to a more complex understanding of how meristems communicate through local and long-distance movement of auxin.
Bennett T, Hines G, van Rongen M, Waldie T, Sawchuk MG, et al. (2016) Connective Auxin Transport in the Shoot Facilitates Communication between Shoot Apices. PLOS Biology 14(4): e1002446. https://doi.org/10.1371/journal.pbio.1002446
Mark Estelle is a Tata Chancellor’s Endowed Professor in Cell and Developmental Biology at the University of California, San Diego, and is a member of the PLOS Biology Editorial Board.
This blog post is the tenth in a series of twelve, forming PLOS Biology’s XV Collection, celebrating 15 years of outstanding open science; read Lauren Richardson’s blog for more information.
Featured image credit: Flickr user NOAA
Mark Estelle image credit: Division of Biological Sciences, UCSD

